Science 1.10
Disinfectants (Investigate life processes and environmental factors that affect them)
Aim:
I am going to investigate how different disinfectant (Dettol) concentrations can affect the growth and reproduction of micro-organisms.
Hypothesis:
I predict that the stronger the disinfectant is, the more bacteria it will kill. I predict this because the active ingredient in Dettol (Chloroxylenol) is effectively used to kill off bacteria by "disrupting the cell wall and stopping the function of enzymes." Wikipedia
Independent Variable:
The independent variable in this experiment is the concentration of the disinfectant. We will change the concentration by diluting the Dettol with different amounts of water. There will be four different concentrations that will be used:
- Full strength (100% Dettol)
- Water (0% Dettol)
- Manufacturer's Instructions (1 capful of Dettol in 400mL water)
- Diluted Dettol (1 capful of Dettol in 800mL water)
The dependent variable in this experiment is the amount of bacteria that is killed. We will measure the clear zone with a ruler, measuring the diameter of the filter paper where the bacteria has been killed by the disinfectant.
- Temperature of environment
This variable will be controlled by putting the agar dish in an incubator and keeping it on the same temperature for the duration of the experiment.
- Size of filter paper
This variable will be controlled by using a hole punch to ensure that all four of the circles of filter paper are exactly the same size.
- Amount of bacterial solution
This variable will be controlled by spreading the bacterial solution evenly over the agar dish by swirling the liquid around, so there is an equal amount in each quarter.
Reliability:
I will ensure that my results are reliable by controlling variables, and by doing the experiment twice, getting data from two other students, and calculating the average.
Equipment:
- Agar plate
- Vivid
- Dropper
- Bacterial solution - peppercorns in water
- Hole punch
- Filter paper
- Tweezers
- Water
- Disinfectant - Dettol
- Beaker
- Cellotape
- Incubator

Method:
- Get out your equipment.
- On the jelly side (inside) of the agar plate, using the vivid, split the plate into four sections.
- Label the sections: full strength, water, manufacturers instructions, and diluted.
- Write your name on the side of the agar plate.
- Using the dropper, transfer some of the bacterial solution onto the agar plate.
- Swirl the agar plate around to cover the whole surface.
- Drain the excess bacterial solution by tipping it into the sink.
- Using a hole punch and some filter paper, punch four disks of filter paper, ensuring they are all the same size.
- Using tweezers, dip one disk into water and shake off the excess, then place it in the middle of one of the four sections on the agar plate. Once it is on the agar plate, don't move it.
- Using tweezers, dip the next disk into the full strength Dettol and shake off the excess, then place it in the middle of the next of the four sections on the agar plate.
- Make up a solution with one capful of Dettol in 400mL of water.
- Using tweezers, dip the next disk into the solution you just made and shake off the excess, then place it in the middle of another one of the four sections on the agar plate.
- Make up a solution of one capful of Dettol in 800mL of water.
- Using tweezers, dip the next disk into this solution and shake off the excess, then place it in the middle of the last of the four sections on the agar plate.
- Cello tape the lid onto the agar plate.
- Place inside the incubator upside down, and leave for two days.
- Observe the results.
- Measure the diameter with a ruler, by measuring right across the clear zone.

Recorded Data (diameter of clear zone):
Water
|
Full Strength
|
Manufacturer's Instructions
|
Diluted
| |
Me - One
|
0mm
|
30mm
|
9mm
|
12mm
|
Me - Two
|
19mm
|
27mm
|
13mm
|
10mm
|
Jorja - One
|
0mm
|
19mm
|
8mm
|
7mm
|
Jorja - Two
|
0mm
|
18mm
|
1mm
|
9mm
|
Te Rama
|
0mm
|
18mm
|
13mm
|
11mm
|
AVERAGE
|
3.8mm
|
22.4mm
|
8.8mm
|
9.8mm
|
Analysis:
Conclusion:
I conclude that my hypothesis was correct - the higher the concentration of disinfection in the solution, the more bacteria it will kill. This was shown in my results, as the full strength Dettol had the highest concentration and killed the most bacteria, while the water had the lowest concentration and killed the least amount of bacteria. My graphs show an increasing trend line, which also proves that the higher concentration killed a larger diameter. There were a few abnormalities, but they ultimately didn’t affect the results too much.
I conclude that my hypothesis was correct - the higher the concentration of disinfection in the solution, the more bacteria it will kill. This was shown in my results, as the full strength Dettol had the highest concentration and killed the most bacteria, while the water had the lowest concentration and killed the least amount of bacteria. My graphs show an increasing trend line, which also proves that the higher concentration killed a larger diameter. There were a few abnormalities, but they ultimately didn’t affect the results too much.
Discussion:
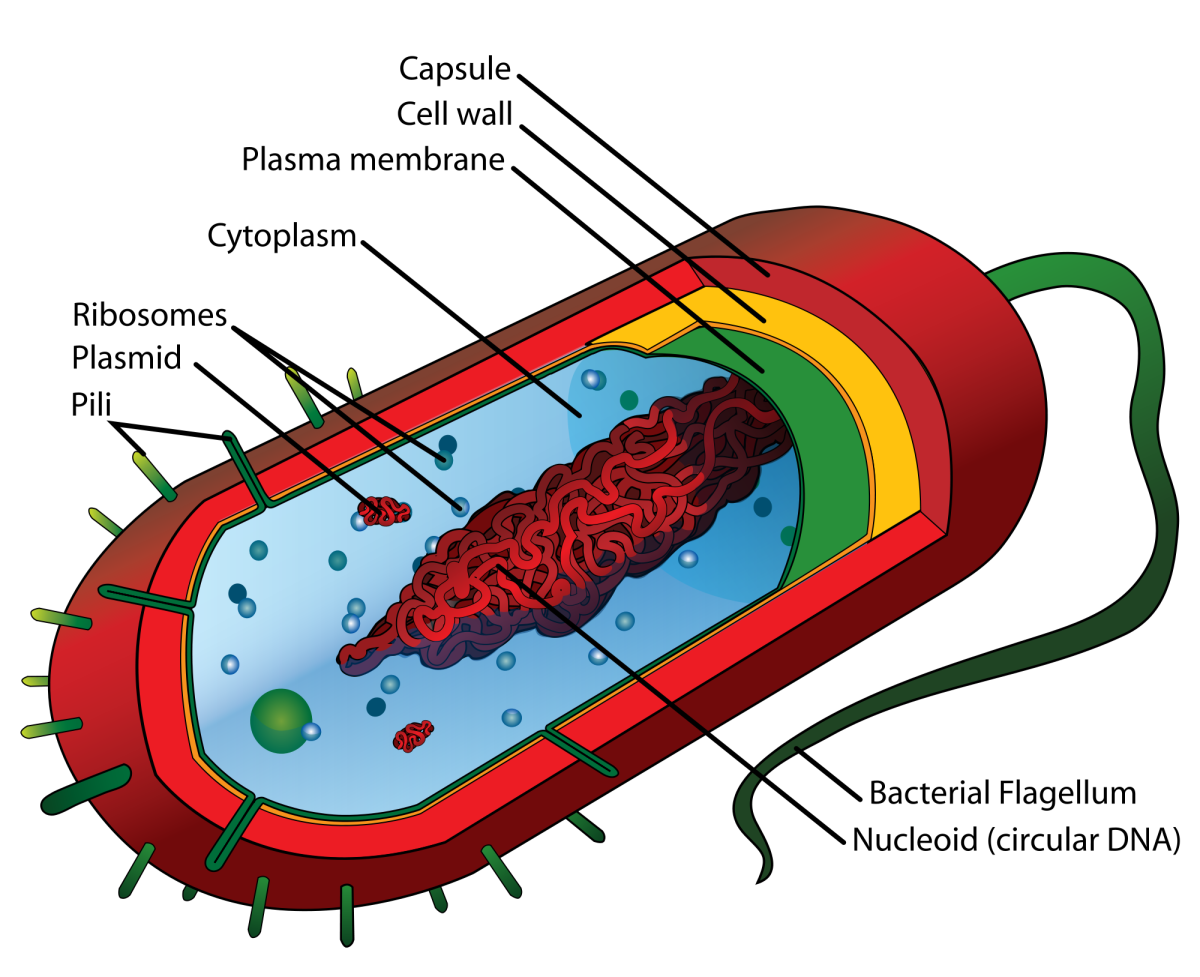
Bacteria are microscopic, unicellular organisms, different to ordinary cells as they have no nucleus. They have a cellular structure made up of the capsule, cell wall, cell membrane, pili, flagellum, cytoplasm, plasmids, ribosomes, and the nucleoid. The capsule is the outer layer of the bacteria. It protects the cell from drying out, and from phagocytosis (engulfment by other cells). The cell wall lies between the capsule and the cell membrane. It gives the cell its shape, and protects the cell membrane. The cell membrane is the last bit of protection the cell has. Its job is to regulate what comes in and out of the cell. Pili live on the capsule. They are small and hairlike, and help bacteria attach to surfaces and other cells. The flagellum is the tail of the cell, and can be located on either end of the bacterium. It moves like a propeller, and pushes the bacterium towards nutrients or away from chemicals. The cytoplasm is the core of the bacterium, a jelly-like substance made up of water, enzymes, nutrients, wastes, and gases. It holds plasmids, ribosomes, and the nucleoid, and is where reproduction and growth of the cell occur. Plasmids are located inside the cytoplasm. They are circular pieces of DNA which give bacteria a selective advantage. Ribosomes are also located inside the cytoplasm, and are where proteins are made. The nucleoid is a section of the cytoplasm, where DNA strands are found.

Bacteria are microscopic, unicellular organisms, different to ordinary cells as they have no nucleus. They have a cellular structure made up of the capsule, cell wall, cell membrane, pili, flagellum, cytoplasm, plasmids, ribosomes, and the nucleoid. The capsule is the outer layer of the bacteria. It protects the cell from drying out, and from phagocytosis (engulfment by other cells). The cell wall lies between the capsule and the cell membrane. It gives the cell its shape, and protects the cell membrane. The cell membrane is the last bit of protection the cell has. Its job is to regulate what comes in and out of the cell. Pili live on the capsule. They are small and hairlike, and help bacteria attach to surfaces and other cells. The flagellum is the tail of the cell, and can be located on either end of the bacterium. It moves like a propeller, and pushes the bacterium towards nutrients or away from chemicals. The cytoplasm is the core of the bacterium, a jelly-like substance made up of water, enzymes, nutrients, wastes, and gases. It holds plasmids, ribosomes, and the nucleoid, and is where reproduction and growth of the cell occur. Plasmids are located inside the cytoplasm. They are circular pieces of DNA which give bacteria a selective advantage. Ribosomes are also located inside the cytoplasm, and are where proteins are made. The nucleoid is a section of the cytoplasm, where DNA strands are found.

In this experiment, we observed the processes of growth, reproduction, and nutrition. Bacteria grows as it is beginning its reproduction process. Bacteria reproduce through a process called binary fission. They do this asexually, and it is done quite quickly. This process creates ‘clones’ of the parent cell. First, the DNA inside the original cell duplicates. Next, the cell begins elongating. Finally, the cell divides in half, and two daughter cells are produced. For bacteria to grow and reproduce, they need nutrients. Bacteria receive nutrients through a process called extracellular digestion. This is where enzymes are let in through the membrane, and are then secreted into food molecules. The enzymes break down the food and are then absorbed by the bacterium. The nutrients are absorbed into the bacterium through diffusion. They diffuse from a high concentration outside the bacterium, through the cell membrane, into a low concentration inside the bacterium. They can also be directly transported into the bacterium, through a pump which acts as a vacuum and sucks the nutrients in.

I predicted that the stronger the disinfectant was, the more bacteria it would kill. I predicted this because the active ingredient in our chosen disinfection was chloroxylenol, which effectively kills bacteria. A disinfectant is a chemical used to kill off bacteria. Our chosen disinfectant was Dettol. We use many different Dettol products in our everyday lives. It can be used as a surface cleaner to get rid of bacteria on benches, or as an antiseptic to clean our wounds. Dettol can eradicate bacteria as it contains the chemical chloroxylenol. This chemical disrupts the cell wall, which means that it can enter the cell membrane as the cell wall protects the membrane. This means that the chemical can filter through into the cell. Once it is inside the cell, it stops the function of enzymes. This means the bacterium cannot absorb nutrients, as enzymes are needed to be secreted into food molecules. If no nutrients are absorbed into the bacterium it cannot grow or reproduce, and therefore is killed. In our experiment we can observe this. We see that the bacteria has stopped growing around the disk, because of the chloroxylenol in the Dettol. It has stopped the bacteria from growing, therefore killing it. The full strength Dettol ultimately killed the most bacteria as it contained the highest amount of chloroxylenol. With this experiment, I now know that I can implement this at home when I am using Dettol to clean surfaces. I don't necessarily need to use full strength, I can also use diluted or manufacturers instructions.
Evaluation:
I can ensure that my results were reliable as I used five different trial dishes. I repeated it myself, used two of Jorja’s trials, and one of Te Rama’s. I then calculated the average. This means that even though there was an abnormality, my results are still reliable. The abnormality was in my second trial. My water disk killed a diameter of 19mm of the bacteria. This is an abnormality as water contains no chemicals, and so no bacteria should have been killed. This could have happened either because I didn’t clean my tweezers each time, or because the water contained chlorine. Next time, I need to sterilize my tweezers before use, and clean them between each solution. I should also use filtered water, as chlorinated water could have an impact on the experiment.
Sources:
https://micro.magnet.fsu.edu/cells/bacteriacell.html
https://en.wikibooks.org/wiki/NCEA_Level_1_Science/Life_processes#Nutrition
https://en.wikipedia.org/wiki/Chloroxylenol